Volume changes during setting of thermoset resins
April 10, 2019Geopolymer Patent Publications
September 17, 2019It is important to consider the thermal properties of thermoset resins when applying them to zonal isolation applications. The key resin properties are the heat of reaction, the rate of reaction, and the coefficient of thermal expansion (CTE).
When resin systems cure significant heat is liberated in a relatively short time which can led to a significant increase in temperature of the resin and surrounding material (casing, cement, rock).
This increase in temperature combined with the fact that the CTE of thermoset resins is much higher (>4x) than that of steel or rock may lead to loss of zonal isolation.
General cases
Take the situation of mismatched thermal expansion coefficients and the effect of temperature changes in annular and plug configurations.
Annular geometry
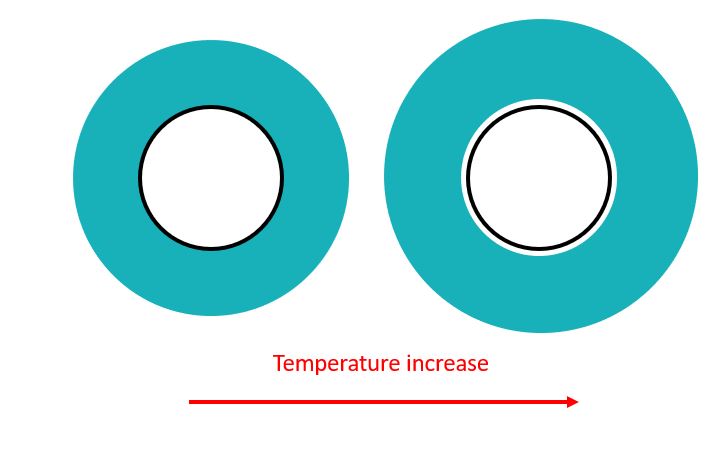
In the picture above the casing is surrounded by a material with a CTE higher than that of the steel. If the temperature is increased, the material will expand away from the casing and create a micro-annulus. However, as discussed in the chemical expansion case the material is confined at its outer surface by another casing string or by rock. If the Young’s modulus of the rock is higher than that of the annular fill material, then the outwards thermal expansion of the material will be limited and consequently no micro-annulus will be formed.
If there is a temperature decrease in this situation then the annular fill material with high CTE will contract onto the casing but will move away from the rock or outer casing. This may lead to generation of an outer micro-annulus.
Plug geometry
With the plug geometry the situation is reversed. With the plug material having a higher CTE than the casing an increase of temperature will cause the material to expand more than the casing, leading to a higher normal force at the material/casing interface and better sealing.
However, if the material and casing are cooled the material will contract away from the casing creating a micro-annulus at the material/casing interface.
Temperature changes during curing or hydration
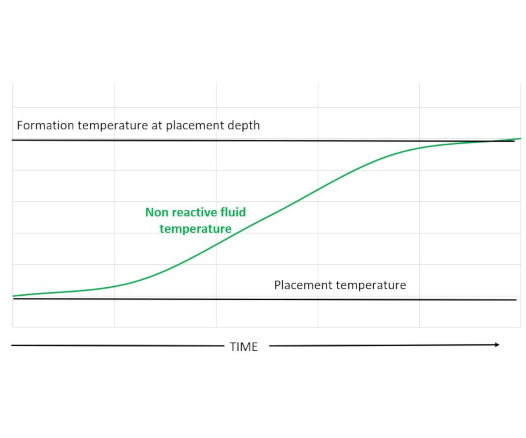
The image at the top of this article shows a temperature profile of a fluid placed within a wellbore as a function of time. The fluid placement temperature is frequently lower than the surrounding formation temperature. After placement the fluid gradually heats up to the formation temperature.
However, if the fluid liberates a significant amount of heat during setting then its temperature and the temperature of the surrounding materials may exceed that of the static formation temperature. This is shown schematically below.
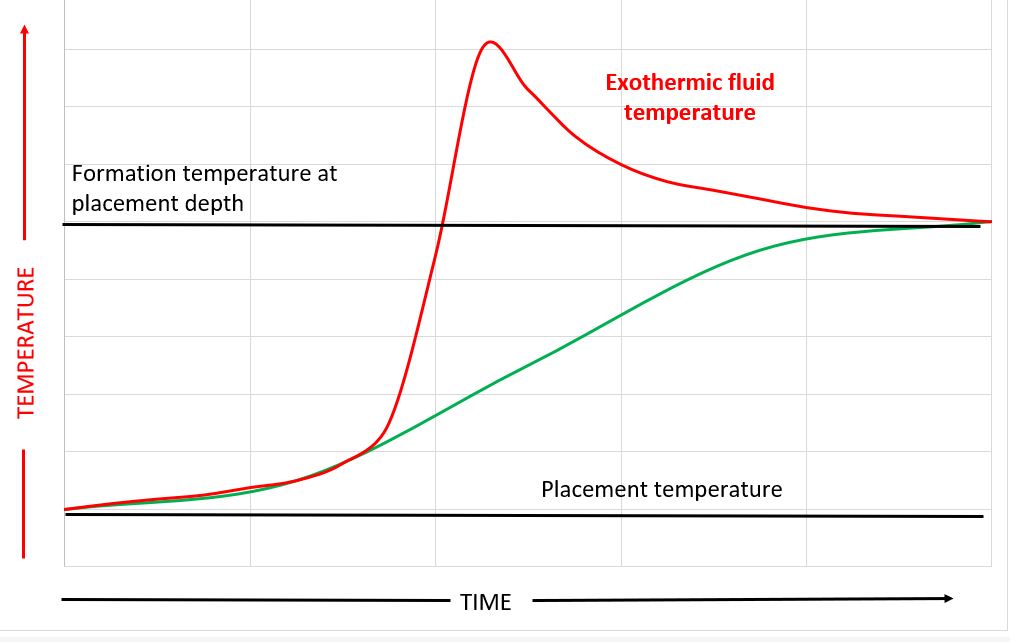
If the material sets (gelation point is reached) at a temperature above the formation temperature then, as explained above, when the material has a higher CTE than the steel or rock, cooling to the formation temperature may result in the formation of a micro-annulus and loss of hydraulic isolation.
Key factors affecting the peak temperature are the volume of the setting fluid and the rate of heat liberation. Large annular volumes and plugs give the highest temperature increases. This situation is recognised in API RP 10B-3, 2nd edition, January 2016, section 7.1, where the temperature during strength testing may be increased if the cement thickness in the annulus >5 cm (effective diameter >10 cm).
Thermoset resins: differences from Portland cement systems
Coefficient of thermal expansion
The CTE of thermoset resins are significantly higher than those of conventional Portland cement systems and of steel. Thus temperature changes have a far greater impact on the performance of resins than that of Portland cement systems.
Some examples:
- A linear CTE of 40 x10-6 °C-1 for an epoxy resin system is given by G.M. Odegard and A. Bandyopadhyay, “Physical aging of epoxy polymers and their composites,” Journal of Polymer Science Part B: Polymer Physics 49(24) 1695‐1716 (2011).
- A linear CTE of 50 to 120 x10-6 °C-1 is given for a vinyl ester resin by Nawab et al., “A device to measure the shrinkage and heat transfers during the curing cycle of thermoset composites,” Advanced Materials Research, Trans Tech Publications, 326, 19-28 (2011).
- US9586863B2 gives a linear CTE of 9 to 10 x10-6 °C-1 for conventional ceent systems and up to 13 x10-6 °C-1 for modified systems.
Heat of reaction and temperature increase
At first sight one might consider that there would be little difference between the temperature increase during curing of thermoset resins and the hydration of Portland cement systems as their heats of reaction are similar.
The heat of hydration of Portland cement is from 320 to 380 J/g. See for example Concrete Technology Today, Volume 18, Number 2, July 1997 from the Portland Cement Association.
The heat of reaction of thermoset resins will depend on the chemistry involved but is in general between 300 and 500 J/g.
- Nawab et al., “A device to measure the shrinkage and heat transfers during the curing cycle of thermoset composites,” Advanced Materials Research, Trans Tech Publications, 326, 19-28 (2011), gives a heat of reaction of 332 J/g for a vinyl ester resin.
- Kim, J., Moon, T. J., & Howell, J. R. “Cure kinetic model, heat of reaction, and glass transition temperature of AS4/3501-6 graphite–epoxy prepregs”, Journal of Composite Materials, 36(21), 2479–2498 (2002), gives a heat of reaction of 475 J/g for an amine cured epoxy resin.
However, the rate of reactions of resins and Portland cement systems can be quite different. The full curing of thermoset resins typically occurs over a few hours once initiated. Portland cement systems take days to fully hydrate. Liberation of a given amount of heat in a shorter time gives a larger temperature increase. This is because there is less time for heat dissipation to the surrounding media.
A comment also has to be made about the mass of reactive material in a given volume. In 1 litre of an 1890 kg m-3 cement slurry there will be approximately 1300 g of cement and the rest will be water (580 g). The density of thermoset resin compositions (without filler) vary from about 1100 kg m-3 to 1400 kg m-3 , so there is little difference in the mass of reactive components in a given volume whether it is a cement system or a resin. However, the specific heat capacity of water is around 3.5 times that of thermoset resins so the temperature increase in the cement slurry will be lower than that of a resin formulation even for the same rate of heat generation.
Setting time and heat liberation
One final point to make is that a significant amount of heat (~60% of the total) is liberated by thermoset resins prior to the gelation point being reached. For Portland cement systems, the equivalent measure is the degree of hydration required for the percolation threshold to be reached. This is a function of the water to cement ratio, but for typical oilfield designs this will be between 20 and 30% (Lavinia et al., “Prediction of elastic properties of cement pastes at early ages,” Computational Materials Science, 47 (3), 775-784 (2010)).
Mitigation of thermal effects
There are several ways to minimise or eliminate issues related to thermal effects during curing of thermoset resins. The key being to minimise the temperature of the resin when the gelation point is reached. If a thermoset resin is used in a plug application, ideally the temperature of the resin should be below the static formation temperature when the gelation point is reached. In this case when the temperature increases to the static temperature the greater thermal expansion of the resin will form a better seal at the resin/casing or resin/formation interface.
Chemistry
The amount of heat generated during curing can be reduced by starting with larger molecules or oligomers. However, the most effective way of minimising the temperature increase is to select a catalyst/crosslinker package that will allow the reaction to occur over many hours, thus giving time for the heat generated to be dissipated.
Fillers
Thermoset resin formulations can include inert fillers such as silica. This reduces the amount of resin in a given volume thereby reducing the heat generated during curing.
Mineral fillers also reduce the coefficient of thermal expansion of thermoset resin formulations (McIlroy, H.M., “Linear thermal expansion of filled epoxy resins,” United States. doi:10.2172/4299891). However, even with large volumes of fillers the CTE is still significantly higher than that of steel or rock.
Placement process
One idea from a patent (US6802375B2 – Shell) is to cool the well significantly prior to placement of a resin. This allows the resin to set at a temperature lower than the static temperature, even considering the exothermic reaction. As the temperature increases to the static temperature expansion of the resin ensures a good seal at the outer interface.